Hydrogen from Biomass
Published on Nov 15, 2016
Abstract
Hydrogen as an energy carrier is considered as one of the solutions to the current energy challenge and represents one of the most promising ways for sustainable energy utilisation. However, renewable, CO2 neutral hydrogen production is very expencive, and most of the hydrogen produced and consumed today is still derived from fossile sources. The technology enables sustainable and efficient production of hydrogen from different biological feedstocks. The Norwegian University of Science and Technology (NTNU) is seeking partners to licence and participate in further development of this novel technology for hydrogen production from biomass.
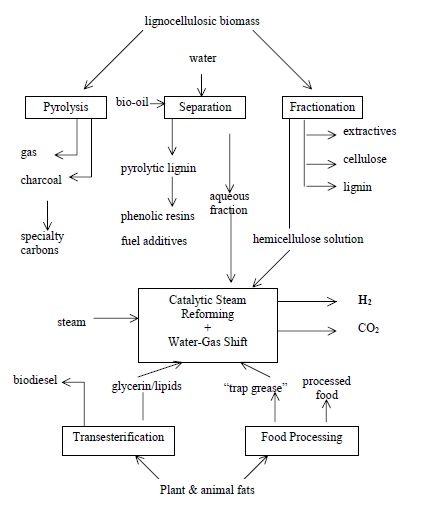
Hydrogen production from biomass is the only direct way to produce hydrogen from renewable energy without a major technology breakthrough. Glycerol is one example of biomass which can be utilised for producing hydrogen. The process opens opportunities to combine biofuel and hydrogen production by using crude glycerol, a by-product, from biodiesel production as feedstock for hydrogen production.
The technology is a one-step method for production of pure hydrogen from biomass by sorption enhanced steam reforming. This technology is wholly owned by NTNU and comprises (i) novel catalysts (ii) novel sorbents, and also (iii) a system for regeneration of the sorption material and catalyst which in all enables continuous production of CO2-neutral and sustainable hydrogen of very high purity (99 % H2 dry basis).
ADVANTAGES
• Sustainable, CO2 neutral hydrogen production
• Production of relatively pure hydrogen (99 % dry basis)
• Higher hydrogen yields in one single step
• Continuous production
• No need for shift reactors or CO2 absorption column
• Only traces of CO
• Lower working temperatures
• Autothermal process
The concepts have successfully been tested in a continous lab scale model on several different biological feedstocks. Nearly 100 % yield of hydrogen with purity over 99 % has been achieved in a single step. Studies on better tolerance using raw bioliquids is the current focus of the research. The process is also highly suitable for other feedstocks besides bioliquids, such as natural gas, multifuels and hydrocarbons in general. The overall goal is to commercialise this technology by involving industrial partners.
Biomass to Hydrogen – Process concept
Materials
Bio-oil used for this study was generated from pine sawdust using the NREL fast pyrolysis vortex reactor system (Diebold and Scahill, 1988). The oil composition (wt%, dry basis) was 47.7% carbon, 7.4% hydrogen, and 44.8% oxygen. Water content was 26.7%. It was separated into aqueous (carbohydrate-derived) and organic (lignin-derived) fractions by adding water to the oil in a weight ratio of 1.5:1. The aqueous fraction contained 20.0 wt.% organics and 80.0% water and consisted of 11.8% carbon, 9.6% hydrogen, and 78.6% oxygen. Natural gas from the public utility had the following composition: 82.4% CH4, 6.6% C2H6, 2.6% CO2, and 8.4% N2 (by volume.)
“Trap grease” was obtained from Pacific Bio-Diesel, a company collaborating with the DOE Bio- Diesel Program. The raw “trap grease” was filtered (at Pacific Bio-Diesel) to remove solid impurities. The grease was a dark-colored liquid of very high viscosity at room temperature. However, at 45°C its viscosity decreased to ca. 80 cP, which made it easy to pump. The grease mainly consisted of fatty acids and their mono-, di-, and triglycerides. The overall elemental analysis of the grease showed 75.5% carbon, 11.8% hydrogen, and 12.7% oxygen. C11-NK, a commercial nickel-based catalyst used for steam reforming of natural gas and naphtha, was obtained from Süd-Chemie (formerly United Catalysts) and ground to a particle size of 300-500µ.
Fluidized bed reformer
The bench-scale fluidized bed reactor is shown in Figure 2. A two-inch-diameter Inconel reactor having a porous metal fluidizing gas distribution plate was placed inside a three-zone electric furnace. The reactor contained 250-300g of commercial nickel-based catalyst ground to the particle size of 300-500µ. The catalyst was fluidized using superheated steam, which is also a reactant in the reforming process. Steam was generated in a boiler and superheated to 750°C before entering the reactor at a flow rate of 2-4 g/min. In the co-reforming experiments, natural gas was compressed and fed to the reactor at a rate of 0.75 L (standard)/min. Liquids were fed at a rate of 2 g/min using a diaphragm pump. In the case of “trap grease,” the feed was preheated and the feeding lines were heat-traced and maintained at 60-80°C to facilitate liquid flow and atomization.
A specially designed injection nozzle supplied with a cooling jacket was used to spray liquids into the catalyst bed. The temperature in the injector was controlled by coolant flow and maintained below the feed boiling point to prevent evaporation of volatile and deposition of nonvolatile components. The product gas passed through a cyclone and hot-gas filter that captured fine catalyst particles and, possibly, char generated in the reactor, then through two heat exchangers that removed excess steam. The condensate was collected in a vessel whose weight was continuously monitored.
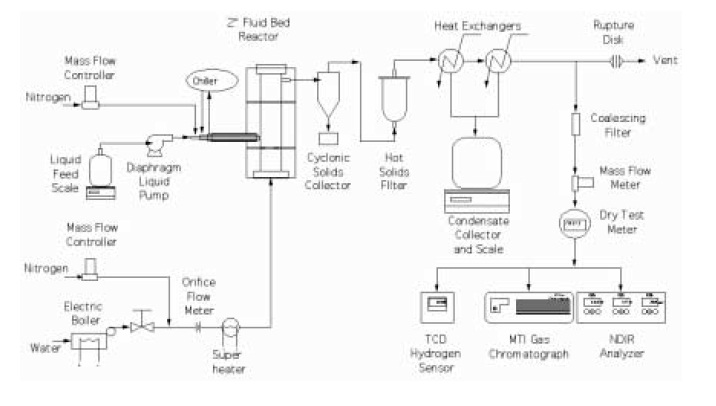
The flow rate of the outlet gas was measured using a mass flow meter and a dry test meter. The concentrations of CO2, CO, and CH4 in the reforming gas composition were monitored by a nondispersive infra-red analyzer (NDIR Model 300) and that of hydrogen by a thermal conductivity monitor (TCM4). In addition, the gas was analyzed every 5 minutes by an on-line MTI gas chromatograph that provided concentrations of hydrogen, carbon monoxide, carbon dioxide, methane, ethylene, and nitrogen as a function of time. The temperatures and flows in the system the flows were recorded and controlled by the OPTO data acquisition and control system. Total and elemental balances were calculated as well as the yield of hydrogen generated from the feed.