Microbivores
Published on Nov 23, 2015
Abstract
Nanomedicine offers the prospect of powerful new tools for the treatment of human diseases and the improvement of human biological systems using molecular nanotechnology. This paper presents a theoretical nanorobot scaling study for artificial mechanical phagocytes of microscopic size, called "microbivores" whose primary function is to destroy microbiological pathogens found in the human bloodstream using a digest and discharge protocol.
The microbivore is an oblate spheroidal nanomedical device measuring 3.4 microns in diameter along its major axis and 2.0 microns in diameter along its minor axis, consisting of 610 billion precisely arranged structural atoms in a gross geometric volume of 12.1 micron.
It is an ideal nanotechnology-based drug delivery system which is—self-powered, computer-controlled medical nanorobot system capable of digitally precise transport, timing, and targeted delivery of pharmaceutical agents to specific cellular and intracellular destinations within the human body. Microbivores will have many applications in nanomedicine such as initiation of apoptosis in cancer cells and direct control of cell signaling process
What would an ideal drug delivery vehicle look like? To start with, it would be targetable not just to specific tissues or organs, but to individual cellular addresses within a tissue or organ. Alternatively, it would be targetable to all individual cells within a given tissue or organ that possessed a particular characteristic (e.g., all cancer cells, or all bacterial cells of a definite species, etc.).
This ideal vehicle would be biocompatible and virtually 100% reliable, with all drug molecules being delivered only to the desired target cells and none being delivered elsewhere so that unwanted side effects are eliminated. The ideal vehicle would remain under the continuous control of the supervising physician, including post-administration. Even after the vehicles had been injected into the body, the doctor would still be able to activate or inactivate them remotely, or alter their mode of action or operational parameters. Once treatment was completed, all of the vehicles could be removed intact from the body, leaving no trace of their presence
Introduction-MICROBIVORE
A nanorobotic device that could safely provide quick and complete eradication of bloodborne pathogens using relatively low doses of devices would be a welcome addition to the physician’s therapeutic armamentarium. Such a machine is the microbivore, an artificial mechanical phagocyte.
The microbivore is an oblate spheroidal nanomedical device consisting of 610 billion precisely arranged structural atoms plus another ~150 billion mostly gas or water molecules when fully loaded. The nanorobot measures 3.4 microns in diameter along its major axis and 2.0 microns in diameter along its minor axis, thus ensuring ready passage through even the narrowest of human capillaries which are ~4 microns in diameter. Its gross geometric volume of 12.1056 micron includes two normally empty internal materials processing chambers totaling 4 micron in displaced volume.
The nanodevice consumes 100-200 pW of continuous power while in operation and can completely digest trapped microbes at a maximum throughput of 2 micron per 30-second cycle, large enough to internalize a single microbe from virtually any major bacteremic species in a single gulp. As in previous designs, to help ensure high reliability the microbivore has tenfold redundancy in all major components, excluding only the largest passive structural elements. The microbivore has a dry mass of 12.2 picograms.
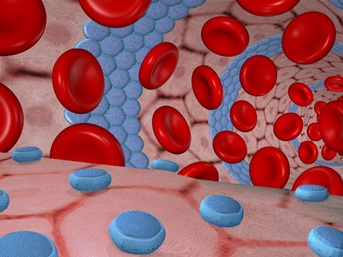
The microbivore, an artificial white cell, floats along in the bloodstream prior to encountering a pathogen. This image, the binding site arrays appear as multicolored circular dapples on the blue sapphire-colored surface
SEPTICEMIA & BACTEREMIA
Septicemia, also known as blood poisoning, is the presence of pathogenic microorganisms in the blood. If allowed to progress, these microorganisms can multiply and cause an overwhelming infection. Bacteremia is the presence of bacteria in the human bloodstream. Although bacterial nutrients are plentiful in blood, the healthy human bloodstream is generally considered a sterile environment. Major antimicrobial defenses include the circulating neutrophils and monocytes (white cells) capable of phagocytosis (engulfing and digesting other cells) and the supporting components of humoral immunity including complement and immunoglobulin’s
Bacteria can enter the blood via injury to the skin, the lining of the mouth or gems, or from gingivitis and other minor infections in the skin and mouth or gums, or from gingivitis and other minor infections in the skin and elsewhere. Bacteria can also enter the blood during surgical, dental, or other medical procedures. Such bacteria are normally removed by circulating leukocytes (along with fixed retculoendothelial phagocytes in the spleen, liver, and lungs), but a few species for disease control estimates that~25,000 U.S. patients die each year from bacterial sepsis. Current therapies often involve multiple antibiotics administered simultaneously in multi-gram quantities per day. These treatments can sometimes take weeks or even months to bring under control certain hardy infectious microorganisms
HOW IT WORKS???
The principal activity which drives microbivore scaling and design is the process of digestion of organic substances, which also has some similarity to the digestion of food. The microbivore digestive system has four fundamental components:
1) An array of reversible binding sites to initially bind and
2) Trap target microbes an array of telescoping grapples to manipulate the microbe, once trapped
3) A morcellation chamber in which the microbe is minced into small, easily digested pieces and 4) a digestion chamber where the small pieces are chemically digested
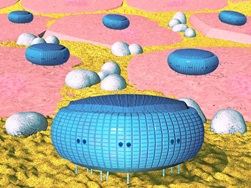
Working of Microbivores
Here’s how the nanorobot works. During each cycle of operation, the target bacterium is bound to the surface of microbivore like a fly on flypaper, via species-specific reversible binding sites. Telescoping robotic grapples emerge from silos in the device surface; establish secure anchorage to the microbe’s plasma membrane, then transport the pathogen to the ingestion port at the front of the device where the pathogen cell is internalized into 2 micron morcellation chamber. After sufficient mechanical mincing, the morcellated remains of the cell are pistoned into a 2 micron digestion chamber where a preprogrammed sequence of 40 engineered enzymes are successively injected and extracted six times, progressively engineered enzymes are successively injected and extracted six times, progressively reducing the morcellate ultimately to monoresidue amino acids, mononucleotides, glycerol, free fatty acids and simple sugars .
These simple molecules are then harmlessly discharged back into the bloodstream through an exhaust port at the rear of the device, completing the 30-second digestion cycle. This “digest and discharge” protocol is conceptually similar to the internalization and digestion process practiced by natural phagocytes, except that the artificial process should be much faster and cleaner. For example, it is well-known that macrophages release biologically active compounds during bacteriophagy, whereas well-designed microbivores need only release biologically inactive effluent.
SENSORS OF MICROBIVORE
The microbivore needs a variety of external and internal sensors to complete its tasks. External sensors include chemical sensors for glucose, oxygen, carbon dioxide, and so forth, up to 10 different molecular species with 100 sensors per molecular species. Each 10 nm × 45 nm × 45 nm chemical concentration sensor with 450 nm2 face area is assumed to discriminate concentration differentials of ~10% and displace ~105 nm3 of internal nanorobot volume .Taking chemical sensor energy cost as ~10 zJ/count with ~104 counts/reading then 10 readings/sec by each of 1000 microbivore sensors gives a maximum sensor power requirement of ~1 pW by a chemical sensor facility that displaces a total of ~0.1 micron3 of device volume and 0.45 micron2 of device surface area.
Acoustic communication sensors mounted within the nanorobot hull permit the microbivore to receive external instructions from the attending physician during the course of in vivo activities. Assuming (21 nm)3 pressure transducers , then 1000 of these transducers displace ~0.01 micron3 of device volume and 0.44 micron2 of device surface area, producing a small net power input to the device of ~10-4 pW when driven by continuous 0.1-atm pulses .
An internal temperature sensor capable of detecting 0.3°C temperature change may have a volume of (~46 nm)3 ~ 10-4 micron3; positioning ten such sensors near each of the 10 independent power plants for redundancy implies a total internal temperature sensor volume of ~0.01 micron3. An additional 0.03 micron3 of unspecified internal sensors are included in the microbivore design, bringing the total for all sensors to 0.15 micron3
Conclusion and Future Scope
Microbivores could also be useful for treating infections of the meninges or the cerebrospinal fluid (CSF) and respiratory diseases involving the presence of bacteria in the lungs or sputum, and could also digest bacterial biofilms. These handy nanorobots could quickly rid the blood of nonbacterial pathogens such as viruses (viremia), fungus cells (fungemia), or parasites (parasitemia). Outside the body, microbivore derivatives could help clean up biohazards, toxic biochemicals or other environmental organic materials spills, as in bioremediation.
References
1) http://www.imm.org/publications/reports/rep025/
2) http://www.kurzweilai.net/meme/frame.html?main=/articles/art0453.html?
3) http://www.rfreitas.com/Nano/Microbivores.htm
4) Soft Machines: Nanotechnology and Life by Richard Anthony Lewis Jones
5) www.nanomedicine.org
6) www.nanorobots.com